Public safety: evolving threats
By Patrick J. Jessee
Lieutenant, Chicago Fire Department, Special Operations, 5-1-2 Hazardous Incident Team Hazardous Materials Training Program Manager, NIPSTA

In its initial inception, the fire service was formed to do exactly what the name describes. It provided protection from just that—fire. Thus, the oath we currently pledge began to take form:
"…protection of property…"
Then the evolution began. Since fire departments were responding to vehicle accidents, it became a marriage of convenience for emergency medical service (EMS) to become integrated into fire protection. Fire stations were staffed 24/7. Fires occurred 24/7. Accidents and medical emergencies happened 24/7. Firefighters began to receive basic emergency medical training such as bandaging, oxygen delivery and CPR. This eventually grew into more advanced life support skills including medicine administration, defibrillation and advanced airway techniques. The oath had to evolve with the job:
"…protection of life and property…"
Quickly the world became more industrialized. Rapid growth meant more chemicals were needed to make machinery work, produce different consumer products, and drive the new industrial world. These chemicals needed to be produced, shipped and consumed by the industry, requiring the transportation of large volumes of chemicals by road, rail, water, air and pipeline. This phase of evolution gave the fire service the new responsibility of responding to hazardous materials incidents. These low-frequency, high-risk events can have dramatic impacts if not handled correctly. Thus the oath, while not formally changing in responsibilities, evolved for hazardous materials teams once again:
"…protection of the environment…"
Then the world began changing with the emergence of terrorism. The weaponization of chemical, biological and radiological materials by global militaries became a threat to the population as well. Already carrying the requisite personnel protective equipment and detection technologies, the fire service found themselves, once again, adopting another responsibility: protecting the population from weapons of mass destruction (WMD). Once again, the fire service found its mission and oath evolving:
"…security and safety of civilians…"
In order for fire department hazardous materials teams to keep up with these increased demands, the hazardous materials community must work with all related parties—fire service, industry, research facilities, manufacturers and military—to approach the complex world of hazardous materials emergency response efficiently and safely. All of these entities must understand each other's goals in order to help develop the best technology and equipment to deliver excellent public safety. This can be an extremely difficult mandate, encompassing organizations with distinctly different personalities and skill sets—from academics who possess significant theories on science and technology, to entry-level recruits with very few experiences to draw from in order to understand these complexities. Producing ruggedized, dependable detection technologies that incorporate sophisticated science and technology while remaining user friendly is crucial for the next evolution of public safety.
Patrick J. Jessee is a lieutenant for the Chicago Fire Department, where he is assigned on 5-1-2, Hazardous Incident Team with Special Operations, stationed at O'Hare Airport. He is also the Hazardous Materials Training Program Manager at Northeastern Illinois Public Safety Training Academy (NIPSTA) in Glenview, IL. Previously, he has completed master's degrees in Public Policy and Administration (Northwestern University), Threat and Response Management (University of Chicago), and Biology (University of Illinois at Urbana Champaign). He also holds bachelor's degrees in Biology (DePaul University) and Chemistry (Southern Illinois University at Edwardsville).
Hazmat through history: Combustible dust
By Daniel Hammel
Marketing Consultant, DuPont Personal Protection

We all know that eating too much sugar can be detrimental to your health, but few of us would consider sugar to be a "hazardous material." However, when that sugar comes in the form of dust, and it's not properly removed from an industrial setting, it could be the cause of a combustible-dust explosion.
Combustible dusts are fine organic or metal particles that present an explosion hazard when in conditions known as the "dust pentagon." The first three elements are known as the "fire triangle": fuel to burn, oxygen and an ignition source. The remaining two elements are a dispersion of dust particles in the right concentration, along with confinement of the dust cloud. A primary explosion may shake loose more accumulated dust, causing a series of secondary explosions that can be far more destructive than the initial explosion. A layer of dust the thickness of a paper clip is potentially hazardous.
At the Imperial Sugar refinery in Port Wentworth, Georgia in 2008, an explosion from ignited sugar dust spread in a chain reaction of secondary explosions within the plant, tragically killing 14 people and injuring dozens more. Flames from the explosions could be seen across the Savannah River in neighboring South Carolina, shooting four stories into the air.
A dust inspection program and proper dust collection systems can help prevent a combustible dust explosion. Poor housekeeping is a major contributor to dust hazards. Cleaning is needed whenever a layer of dust reaches 1/32 inch over 5% of the surface area, including beams, ducts and equipment. It's imperative that cleaning methods do not generate dust clouds if ignition sources are present.
To find out more about DuPont protective apparel for industrial cleaning, please visit www.SafeSPEC.DuPont.com
Chemical protective clothing selection 101: Finding the best match starts with your hazard assessment
By Susan Lovasic
Global Regulatory Affairs Manager, DuPont Personal Protection
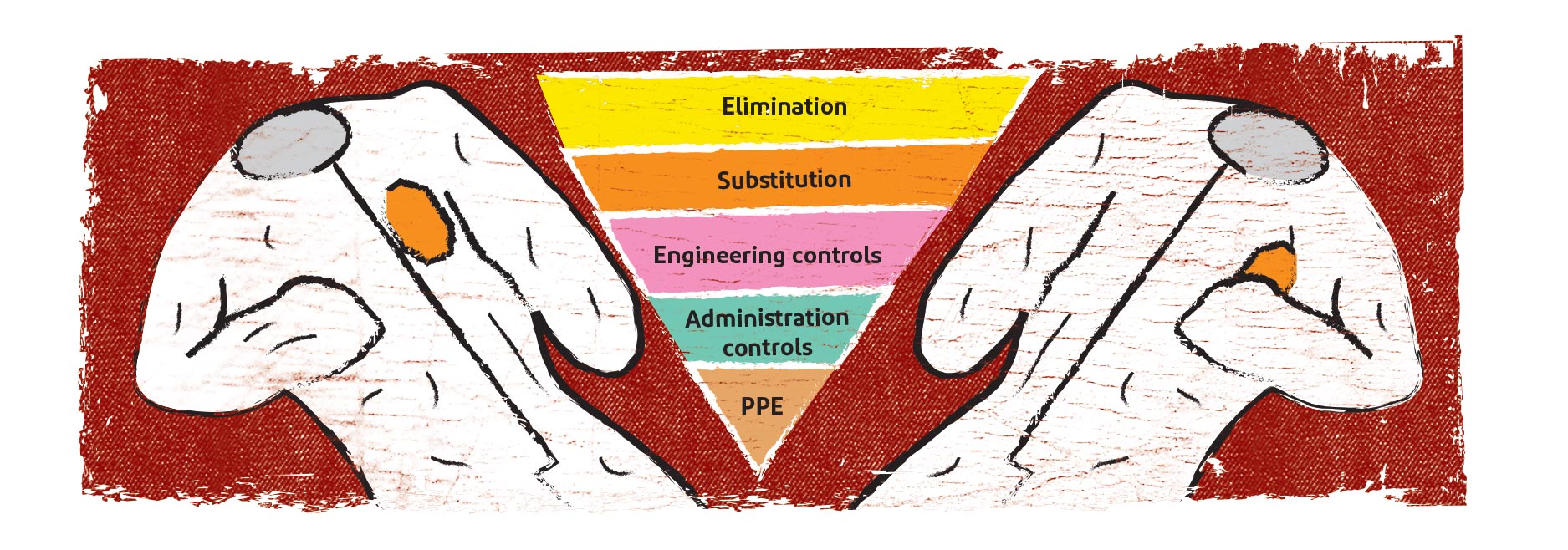
Safety professionals deal with many workplace hazards. In order to help reduce or eliminate these hazards, most employ the "Hierarchy of Controls" shown above. If possible, eliminate the hazard completely or substitute a safer option. If these options are not possible, apply engineering controls to minimize worker contact. If that is not 100% effective, administrative controls such as work practices, should be modified to minimize potential contact. Only after all of these steps are completed should personal protective equipment (PPE) options be selected for any residual risk. Employer assessment of workplace hazards drives the selection of appropriate PPE.
Hazard assessments
In order to properly select chemical protective clothing (CPC), there are several questions that should be considered during the hazard assessment. First, what is the hazardous chemical's name, Chemical Abstracts Service (CAS) Registry number, and the chemical's physical state (solid, liquid or gas)? Many industries use their own chemical names or acronyms. It is best to use the CAS number to ensure that you are referring to the same chemical as a CPC manufacturer. Is the chemical a mixture? What are the components? A hazardous component could breach CPC barrier material as part of a mixture, but not while in its pure form.
Secondly, how is the chemical a skin hazard? Will it cause skin irritation or burn? Can the chemical be absorbed through the skin and cause acute or chronic health effects? It is important to consider that some materials are not listed as skin hazards, but if these particles get onto a worker's skin or clothing, they can be transferred and become inhalation or ingestion hazards at the workplace or after leaving.
Finally, consider the potential exposure scenario. This is best done by directly observing the work. What are the work practices? Is chemical exposure expected to occur frequently or only when something fails in the process? Could a specific non-routine work task (e.g., line break) cause exposure? Consider how much of the chemical could be released and which direction it could move. The exposure scenario should consider each of these factors: likelihood of contact with the chemical, amount, duration and direction.
Once the chemical hazard assessment has been completed, then CPC selection can begin. Remember that CPC is often an element of a protective ensemble. You must validate that the wearer is fully protected from all hazards, including respiratory hazards. CPC selection, along with the specific barrier properties of fabric used in CPC, will be addressed in the next edition of The Glow Worm. Stay tuned!
If you'd like to learn more, the full version of this article originally appeared in the May 2019 issue of Occupational Health & Safety.
Protection in an uncontrolled environment: Emergency responders & opioids exposure
By Mark Tartaglia, MSPH, CIH, DuPont Personal Protection
and Robert N. Phalen, PhD, CIH, University of Houston-Clear Lake

The full version of this article originally appeared in the December 2017 issue of The Synergist®, the magazine of the American Industrial Hygiene Association®. The shortened version below is reprinted with the permission of AIHA.
Opioids are an important class of drugs prescribed to patients to help manage chronic pain. When legally prescribed and taken as directed, opioids are very effective at pain management. Unfortunately, they are highly addictive and subject to abuse. Overexposure or abuse of these drugs can cause respiratory arrest and death.
Anticipating emergency responder exposures
Synthetic opioids come in a variety of physical forms including pills, powders, liquids, nasal sprays, dermal patches and on blotter paper. Different levels of risk are associated with the route of exposure and amount. Powders are a concern because they can easily become aerosolized and inhaled, especially when opening a container to check its contents. Liquids can also be of concern due to the potential for absorption via the skin, eyes, and mucous membranes, or vaporization upon heating that occurs in a fire. Common exposure scenarios for emergency responders include the following:
Contact with drug users. Law enforcement officers and EMS personnel can be exposed to drug users carrying small amounts of synthetic opioids when responding to a drug overdose or death, or when searching a suspect. The threat may not be readily visible under these circumstances, but exposure risk is typically lower when the drug is contained and in smaller amounts.
Collection of evidence and on-site testing. Exposures can occur when officers or agents collect suspected opioids as evidence or test them on site (for example, using colorimetric indicators). Opening containers or baggies is likely to release the powder into the air. Many law enforcement agencies now have policies that discourage this practice in an uncontrolled environment.
Distribution and production. Drug raids and investigations of production or distribution locations are more likely to result in higher exposures. According to a May 15, 2017 report in The Atlantic, flashbang grenades—nonlethal weapons intended to stun adversaries—can aerosolize large amounts of liquid or powder opioids, which has resulted in significant adverse health effects in law enforcement personnel.
Fires. Structural fires pose an additional risk on top of the common thermal and physical hazards. Both inhalation and dermal contact with opioids may exist. Synthetic opioids are water-soluble and thus pose less of a hazard if saturated with copious amounts of water. However, dermal contact with heavily contaminated water could pose a hazard for dermal absorption.
Recognizing and evaluating risk
Law enforcement and other public safety workers may have to contend with a variety of potential exposure scenarios that present varying levels of risk. Those with high potential exposures include special operation squads raiding active clandestine labs and "pill mills", as well as investigative personnel collecting evidence. Moderate risk may be associated with routine law enforcement or emergency medical actions where visible and suspect powder material is present. Low risk may be related to law enforcement or medical response scenarios where illicit drug activity is suspected, but no visible powders are present. General safety precautions, such as disposable gloves and a uniform that covers the skin, may afford protection against incidental contact; but these will not be effective against larger exposure.
Fentanyl and its derivatives, as well as laced illicit drugs, typically are encountered in powder form, which presents both inhalation and dermal exposure risks. Due to the high bioavailability of these opioid particles, the greatest exposure risk is attributable to inhalation or mucous membrane exposures. Despite this, dermal contact poses a secondary exposure risk that must be protected against, especially if the skin is moist.
For these reasons, emergency response personnel must be trained to anticipate and recognize situations where an exposure to opioids may exist and then assess the level of associated risk.
Indicators of exposure
The first step to recognizing a hazard is to establish clear indicators. The primary indicator of an opioid overdose is respiratory depression or arrest. The more objective signs of an overdose include slowed breathing, cessation of breathing, lack of oxygen with blue discoloration of the lips and skin, and possibly a depressed level of consciousness. Secondary signs, such as pinpoint pupils, clammy skin and drowsiness, may also be associated with opioid use or overdose. More generalized signs of dizziness, nausea, or anxiety are not reliable indicators of an overdose. The presence of drug paraphernalia and powders in the vicinity can also lead to an assumption of possible illicit drug use. Distribution and production facilities are likely to have chemical synthesis equipment, weighing scales, pill presses and packaging materials present.
Levels of risk
Several groups have developed guidelines to assist with performing related risk assessments. Both NIOSH and the InterAgency Board (IAB) have established risk levels based on the exposure scenario. These risk levels are then used to recommend effective PPE and controls.
Exposure controls
Minimizing exposures in emergency responders requires a focused and structured effort that aims to prepare personnel to anticipate, recognize and assess risks. This approach involves:
- Training responders on the proper selection, care, use, decontamination and storage of PPE
- Training responders to recognize and assess hazardous scenarios
- Providing levels of PPE protection appropriate for the anticipated levels of exposure
- Training responders on the proper donning and doffing of PPE to control exposures
- Establishing standard operating procedures for exposure control and chemical hygiene
- Training responders to recognize the signs and symptoms of opioid intoxication
- Training responders on how to wash opioids from the skin, eyes, nose and mouth
- Providing naloxone injectors to reverse the lethal effects of opioids
- Training responders on the administration of naloxone to affected patients or responders
Personal protective equipment
Since many of the exposure scenarios are uncontrolled situations, it is often difficult to use engineering controls, such as ventilation, to capture aerosolized powders. Consequently, protecting responders depends heavily on PPE.
Respiratory protection. For moderate exposure risks, a NIOSH-approved N95 or P100 respirator is typically recommended. Higher levels of respiratory protection, such as a powered-air purifying respirator (PAPR) or self-contained breathing apparatus (SCBA), may be warranted under more severe exposure conditions.
Eye and mucus membrane protection. Both eye and face protection are recommended when the possibility of facial contact exists. This is because fentanyl and related opioids cross mucous membranes at a higher rate than through the skin. Similar to inhalation, exposure through the eyes, nose or mouth could lead to more acute and severe toxicity.
Dermal protection. Uniforms and disposable nitrile gloves have been shown to provide adequate dermal protection against incidental exposures. Larger exposures may require double gloving or use of thicker nitrile gloves, as well as a water-resistant, taped-seam coverall or encapsulated suit.
Incidental dermal exposures need to be immediately washed away with copious amounts of water plus a mild soap or detergent, if available. Alcohol-based hand sanitizers and hypochlorite bleach solutions should be avoided, as they may not effectively dissolve or remove opioids from the skin. Alcohol-based products may increase dermal absorption.
The keys to protecting emergency responders from potent and legal synthetic opioids are to anticipate exposures, recognize and evaluate risks early on, provide adequate PPE, and have well-established hygiene and control procedures that trained workers can implement. The level of control and PPE depend on the route of exposure and the amount of exposure.
Products and/or sales questions?
Share your stories, tips and tricks.

Resource library
Find technical information, videos, webinars and case studies about DuPont PPE here.


