Enhancing advanced PCB performance with corrosion-resistant electroless nickel
May 23, 2016
Printed circuit boards (PCBs) are shipped to assemblers with a “final finish,” a solderable surface that provides excellent electrical and mechanical connections to IC packages and other components attached to the bare board. The most common final finishes today are Organic Solder Preservatives (OSP), electroless nickel/immersion gold (ENIG), immersion tin and immersion silver. In this discussion, we will review options related to the ENIG process.
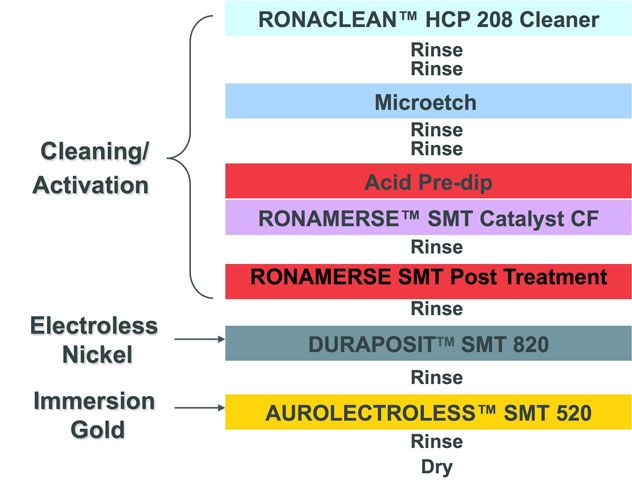
Figure 1: ENIG Process Flow
Figure 1 shows the standard ENIG process flow involving cleaning/activation of copper surfaces, followed by electroless nickel (EN) and immersion gold (IG). As its name suggests, the EN is chemically reduced, using hypophosphite ions (H2PO2-). As a byproduct of this reaction, some phosphorous co-deposits with the nickel—typically about 6-9% (by weight)—provide corrosion resistance to this deposit. The subsequent IG process is a displacement process: Gold is deposited onto the surface as nickel is dissolved into the IG solution. This inherent corrosion of nickel is something that must be taken into consideration when formulating ENIG chemistries.
Selective ENIG
Becoming more common in PCB production are mixed finish requirements, especially selective ENIG (S-ENIG). An S-ENIG process deposits ENIG only on the features that will require wire bonding, or that will serve as touch pads or contacts, with OSP deposited on the remaining solderable features. The potential benefits of this finish are the reduction of gold usage and lower cost. The general process flow is as follows:
- Photo-masking features that will eventually be finished with OSP using dry-film
- ENIG plating of exposed copper
- Stripping dry-film
- OSP coating (cleaner > microetch > OSP with rinses between steps)
An inherent step in the OSP process is a microetch, which ensures that the copper surface is clean prior to coating. However, the microetch can attack the nickel, depending on process conditions. Because of this, and for determination of process capability, most S-ENIG processes are evaluated for nickel corrosion after two passes through OSP in case there is a need to rework the OSP. One way to minimize or prevent this condition is to use a high-phosphorous (“phos”)-content EN (typically 10-12% by weight)—as compared to a standard 7-9% mid-phos EN, to provide increased corrosion resistance.
As detailed in Figure 1, Dow Electronic Solutions has long supplied a synergistic family of formulations to allow our customers to create optimized ENIG final finishes for different applications. Dow’s Duraposit™ SMT820 EN is formulated to produce a deposit with ~10-11% phosphorous by weight, providing a nickel deposit for optimum corrosion protection when integrated with Aurolectroless™ SMT 520 IG. SMT 520 IG enables low gold concentration in the IG bath (0.5-0.7 g/Lt) to reach gold thickness of 1.2~2.5µm, and provides a high tolerance of contaminants for a long bath life.

Figure 2: FIB Analysis of Ni Corrosion Resistance
An example of the difference in corrosion resistance may be seen in Figure 2, where the high-phos EN performs better than the mid-phos EN. Additional testing of the high-phos SMT 820 EN has encompassed corrosion resistance performance to multiple passes through OSP (Figures 3a, b and c), resistance to SO2 gas corrosion, and ball-shear testing for solderability (Figure 4). In all cases, the ENIG finish using high-phos EN met all specifications.

Figure 3a: Corrosion resistance performance (via) during OSP

Figure 3b: Corrosion resistance performance (hole) during OSP
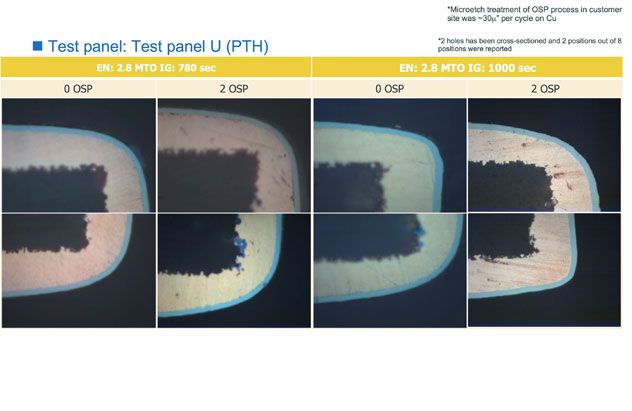
Figure 3c: Cross section result of corrosion resistance performance during OSP
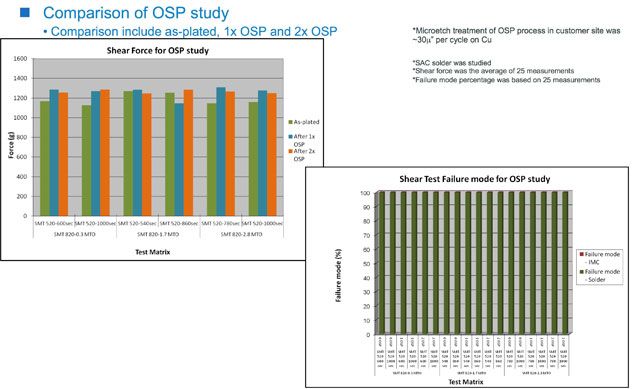
Figure 4: Normal ball shear test for solderability following OSP
Duraposit SMT 820 EN adds a high-phos option to Dow’s final-finish product portfolio, allowing the PCB fabricator choices for different applications. Whether it’s for an S-ENIG finish or for a high-reliability application, SMT 820 provides high corrosion-resistance performance to help meet the needs of the most advanced PCBs powering a myriad of today’s electronic devices.
Related Information

DuPont keeps up with trends in integrated circuit (IC) substrates and understands the need for cost-effective solutions that enhance package reliability.

The phosphorous content of the electroless nickel deposit improves the corrosion resistance of the ENIG deposit, particularly for selective gold processes using OSP coatings

High-performance chipsets for today's routers, switches and servers calls for advanced Cu plating.
We’re here to help.
We love to talk about how our electronics solutions can build business, commercialize products,
and solve the challenges of our time.